Magnesium alloys are widely used due to their excellent mechanical strength, creep resistance, damping performance and heat-conducting property.
However, the activity of the magnesium element can cause the electrochemical corrosion of magnesium alloys, especially in the marine atmospheric environment.
Recently, a research team led by Prof. Hou Baorong from the Institute of Oceanology of the Chinese Academy of Sciences (IOCAS) studied the corrosion behavior of Mg-Y-Nd-Zn-Zr magnesium alloys in the South China Sea.
The study was published in Journal of Magnesium and Alloys on May 5.
The researchers analyzed the corrosion resistance of different alloys via weight loss rate and electrochemical measurement in the laboratory. They found that Mg-5Y-1.5Nd-xZn-0.5Zr magnesium alloy exhibited the best corrosion resistance.
The resistance for corrosion of magnesium alloys in the typical marine environment was as followed: Mg-5Y-1.5Nd-6Zn-0.5Zr < Mg-5Y-1.5Nd-0.5Zr < Mg-5Y-1.5Nd-2Zn-0.5Zr < Mg-5Y-1.5Nd-4Zn-0.5Zr.
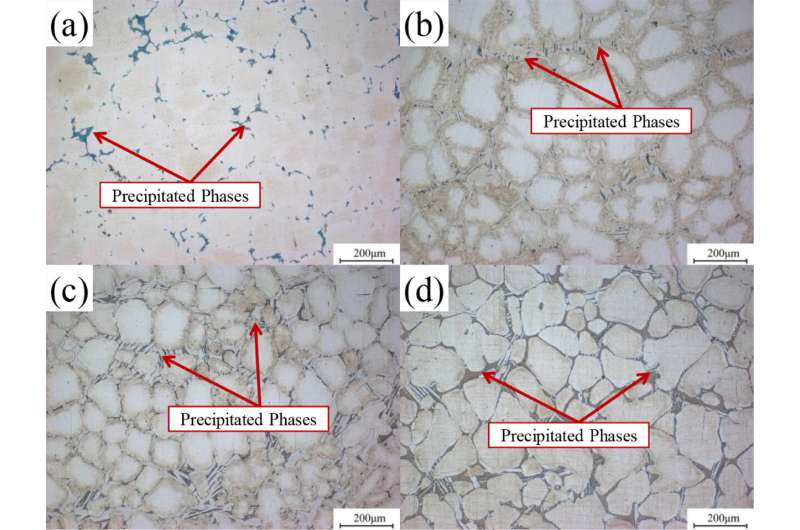
The researchers investigated the microstructure of the alloys in order to explain the corrosion mechanism. The standard electrode potential of rare earth elements was more negative than that of pure magnesium. The precipitated phases in the magnesium alloys containing rare earth elements could influence the micro-anode.
“Some special anodic precipitated phases exhibited biaxial synergistic effect, micro-galvanic corrosion acceleration and corrosion barrier. This can improve the corrosion resistance of magnesium matrix to some extent,” said Jiang Quantong, first author of the study.
“Regulating the microstructure, type and distribution of precipitated phases in magnesium alloys containing rare earth to improve both the mechanical strength and corrosion resistance needs further study,” said Prof. Hou.
You may like to read:
Mg-Li Battery-Suitable Highly Dispersed MoS2 Nanoflowers Created
Computational sleuthing confirms first 3D quantum spin liquid